The Science of Soil Analysis
Soil analysis can be broken down into 3 main factors: Soil pH, soil color, and soil conditions. Optimizing any or all of these factors by using MultiFIX® will increase your soil’s overall health, leading to higher crop yields and quality.
Let’s take a closer look at each of these factors and how they can affect your crops.
Soil Analysis and pH
pH stands for “potential Hydrogen” and is a way of measuring the acidity or alkalinity in your soil. Having the correct pH is important for healthy plant growth. Being aware of the long-term effects of different soil management practices on soil pH is also important, because some agricultural practices significantly alter soil pH.
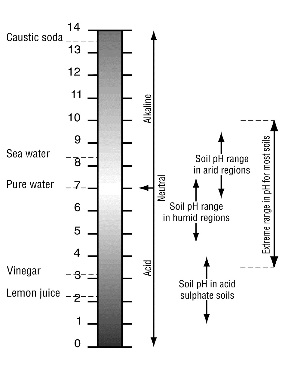
Since pH value is actually a measure of hydrogen ion concentration, and because hydrogen ion concentration varies over a wide range, a logarithmic scale is used. For every pH decrease of 1, the acidity increases by a factor of 10.
Most soils have pH values between 3.5 and 10. In wetter areas the natural pH typically ranges from 5 to 7, while in drier areas the range is 6.5 to 9.
Soils can be classified according to their pH value:
- 6.5 to 7.5—neutral
- over 7.5—alkaline
- less than 6.5—acidic, and soils with pH less than 5.5 are considered strongly acidic.
Most plants do well in soil with a pH of 6.5, slightly acid. (see pH Values) . However, rhododendron, camellias, azaleas, blueberries, ferns, spruce, pines, firs, and red cedar prefer soil with a pH of 4.0 to 5.0. Pines, firs, holly, daphne, spruce oak, birch, willow, rhododendron, alder, and red cedar grow well in soil with a 5.0 to 6.0 pH. Soils with a pH of 6.0 to 7.0 will grow maple, mountain ash, pansy, asters, peaches, carrots, lettuce, pines, firs, alder, and red cedar. Beech, mock orange, asparagus and sagebrush tolerate soils with a pH 7.0 to 8.0. Above 8.5 the soil is too alkaline for most plants and soil with a pH less than 3.5 is too acid. Each soil layer may have a different pH.
To determine soil pH, a universal indicator or pH paper can be used. Put a small amount of the soil to be tested in a clear or white container. Do not touch the sample. Pour a small amount of universal indicator over the soil. Match the color of the indicator with the pH color chart. If using indicator paper, pour a small amount of water on the soil sample. Touch the indicator paper to the sample and match to color of the paper to the pH color chart.
Soil pH can be raised, making the soil less acidic and more alkaline, by adding lime to the soil.
Soil Color
Soil color gives information about organic matter in the soil, drainage, biotic activity and fertility.
| Condition | Dark (dark grey, brown to black) | moderately dark (brown to yellow brown) | light (pale brown, yellow) |
|---|---|---|---|
| organic matter | high | medium | low |
| erosion factor | low | medium | high |
| aeration | high | medium | low |
| Available nitrogen | high | medium | low |
| fertility | high | medium | low |
Condition | Subsurface soil color |
|---|---|
water-logged soils, poor aeration | dull grey (if in low rainfall soils 0-20 in.) |
well drained soils | yellow, red-brown, black (if in forest soils) |
somewhat poorly drained soils | mottled grey (if in humid soils) |
Soil Conditions
The organic content of soil greatly influences the plant, animal and microorganism populations in that soil. Decomposing organic material provides many necessary nutrients to soil inhabitants. Without fresh additions of organic matter from time to time, the soil becomes deficient in some nutrients and soil populations decrease. The amount of organic material can be determined by ignition. Organic material is made of carbon compounds, which when heated to high temperatures are converted to carbon dioxide and water. In the ignition process, a dry solid sample is heated to a high temperature. The organic matter in the soil is given off as gases. This results in a change in weight which allows for calculation of the organic content of the sample.
Oven-dry the sample to remove water (see soil moisture). Weigh a crucible and lid, evaporating dish and cover, or other covered container. Place approximately 10 grams of soil sample in the container, cover it and weigh the sample, container and cover. Place the container on a metal stand and heat it with a propane torch. Allow the fumes to escape, but not the soil particles. Heat the sample strongly after most of the gases have escaped; continue heating until there are no visible fumes. Cool the container, lid, and sample. Reweigh and calculate the percent of organic material.